Passiflora auriculata
P. auriculata is one of the two most common species of Passiflora in second growth and edge habitats at La Selva. The large yellow nectaries at the base of the leaf petiole and on the leaf blades are unique, and are very attractive to ants. P. auriculata plants frequently sprawl with many short branches but larger vines may be present with thick triangular or sometimes flattened stems. P. auriculata flowers are fairly small as are the dark purple or black fruits. The nectaries on the leaf blade may be distinctly yellow and may act as egg mimics to deter oviposition by Heliconius butterflies. Range: Nicaragua to Bolivia, Brazil and Venezuela. Wet forest from sea level to 1700 meters elevation. A member of section Auriculata, subgenus Decaloba, with principal diversity in the Amazon basin.
HCN measurements reveal that P. auriculata foliage produces small but variable amounts of HCN when crushed (0.01 to 1.0 μM HCN per gram of new leaves), with tiny seedling leaves ranging up to 10 μΜ/g. P. auriculata contains the simple monoglycoside cyclopentyl cyanogen Epivolkenin, with tiny amounts of other cyanogens present (Engler et al 2000). The 2 orders of magnitude range appears to be significant to many of the flea beetle species, and most plants with significant flea beetle feeding damage are at the lower end of the range. Rootlets, like seedling leaves, have 10 times larger amounts, ranging from .6 to 6 μM/g HCN (wet weight).
I investigated P. auriculata HCN variation in more detail by locating 11 widely spaced individual plants, each with multiple branches along a common stem. I collected three leaves from each branch, including a new leaf not yet fully expanded, a full-sized new leaf, and the oldest leaf. In 5 cases I could verify the common origin of each branch by tracing the stems downward and locating the connection, but in 6 other cases the stems ran under logs and debris and could not be followed. In those cases I judged that the spatial separation between plants was sufficient that I could assume a common connection. I did not use leaf appearance, maturity, or degree of shading as criteria for identifying connected branches. The results of over 100 measurements expressed on a log scale may be seen in Figure 5a. Within-species variation in HCN. The figure reveals average within-branch variation of about 0.7 log units (5x), between-branch variation of about .4 (2x), and a between-plant variation of about 1.0 (10x). See Figure 5d and charts below for more details. About half of the 100-fold variation in P. auriculata leaf HCN may be attributed to differences between plants, and the other half to variation among branches and leaves within a plant. It is interesting to note that I could not see any external differences between high- and low-HCN plants, other than the amount of flea beetle feeding damage. The latter was often inversely correlated with quantity of HCN (see chart below). Measurements of cyanogenesis in P. auriculata may also be seen in charts in Appendix 2: charts A4, A5 and A6.
H. cydno, H. hecale and especially the specialist H. sara lay their eggs on P. auriculata at La Selva, the former species on the tendril tips and the latter laying her clutches of eggs on the shoot tips among the incipient leaves. If H. sara clutches escape parasitism by egg parasites (Trichogrammatidae and Scelionidae) and social wasps, the resulting 30-50 caterpillars can rapidly defoliate a plant. Measurements show that the larvae of the P. auriculata specialist H. sara do not release any detectable HCN while feeding on this plant.
This species is also eaten by several species of flea beetle, including the generalist Blue Flea Beetle (Monomacra violacea), the Yellow-legged Flea Beetle (Parchicola DF-2), and the Red-brown-white Flea Beetle (Ptocadica bifasciata). The black-tibia and the yellow tibia Parchicola species have also been observed on several occasions. The Striped Flea Beetle (Disonycha quinquelinata) was observed once on this plant. Pt. bifasciata eggs and larvae have been found on the leaves of the plant, as have the immature stages of Parchicola DF2. Pa. DF2 eggs are laid at the base of the plant amoung the surface rootlets or, alternatively, on the underside of leaves near the base of the plant. Flea beetles often "infest" individual P. auriculata plants for long periods (perhaps permanently) once a large patch of plant is found, but (unlike P. lobata) I have seldom seen patches die off or otherwise disappear. Perhaps the elevated amounts of cyanogenic glycosides provide some protection for the roots and stems.
I have confined M. violacea adults on P. auriculata in cages in the shadehouse, but no reproduction has occurred.





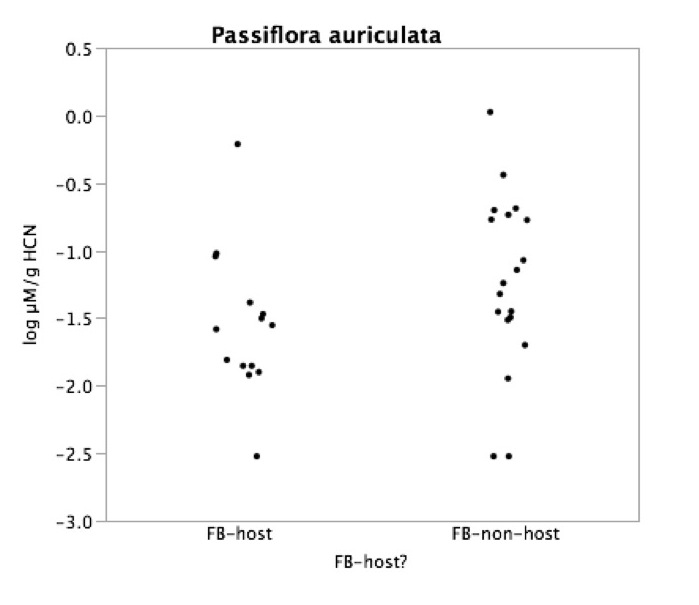
Right: Passiflora auriculata plants with substantial flea beetle feeding damage vs. those without damage: Comparison of HCN content in foliage. Note non-significant trend (p=.16) for flea beetle host plants to have reduced amounts of HCN production (about 50% less). HCN was measured from a single leaf on each plant.
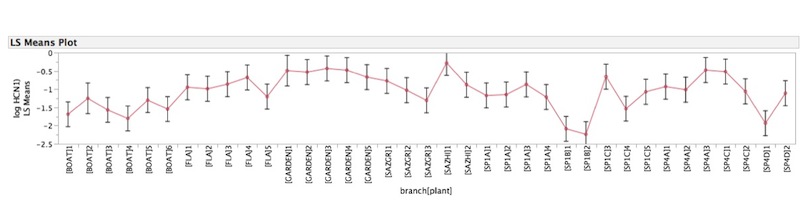
Below: Analysis of variance of HCN produced by crushing 3 leaves from 37 branches of of P. auriculata (log μM/g). The 37 branches were taken from 11 individual plants. This graph shows the average HCN per plant and the standard errors of the means. Plants vary significantly in average μM/g HCN (ANOVA p<0.0001)

Mean quantity (μM/g) of HCN produced by crushing 3 leaves from 37 branches of of P. auriculata. The 37 branches were taken from 11 individual plants. The bars represent standard error of the mean. After subtracting the average differences among plants, the remaining within-plant variation between branches is also significant (p=0.002).
On average I found no difference in HCN content between new (left) and older leaves (right) for P. auriculata.
